 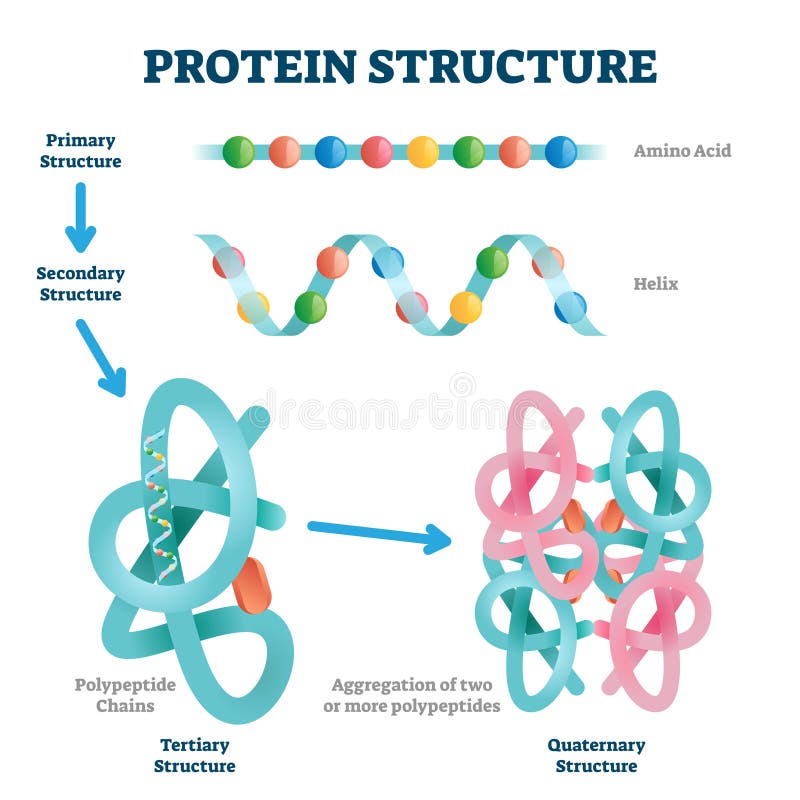 
This is because certain amino acids with sulfhydryl or SH groups form disulfide (S-S) bonds with other amino acids in the same chain. In globular proteins such as enzymes, the long chain of amino acids becomes folded into a three-dimensional functional shape or tertiary structure.The secondary structure of the protein is due to hydrogen bonds that form between the oxygen atom of one amino acid and the nitrogen atom of another and gives the protein or polypeptide the two-dimensional form of an alpha-helix or a beta-pleated sheet.Since certain amino acids can interact with other amino acids in the same protein, this primary structure ultimately determines the final shape and therefore the chemical and physical properties of the protein. The order of deoxyribonucleotide bases in a gene determines the amino acid sequence of a particular protein.The actual order of the amino acids in the protein is called its primary structure and is determined by DNA.Therefore, proteins are long chains of amino acids held together by peptide bonds. A peptide is two or more amino acids joined together by peptide bonds a polypeptide is a chain of many amino acids and a protein contains one or more polypeptides. 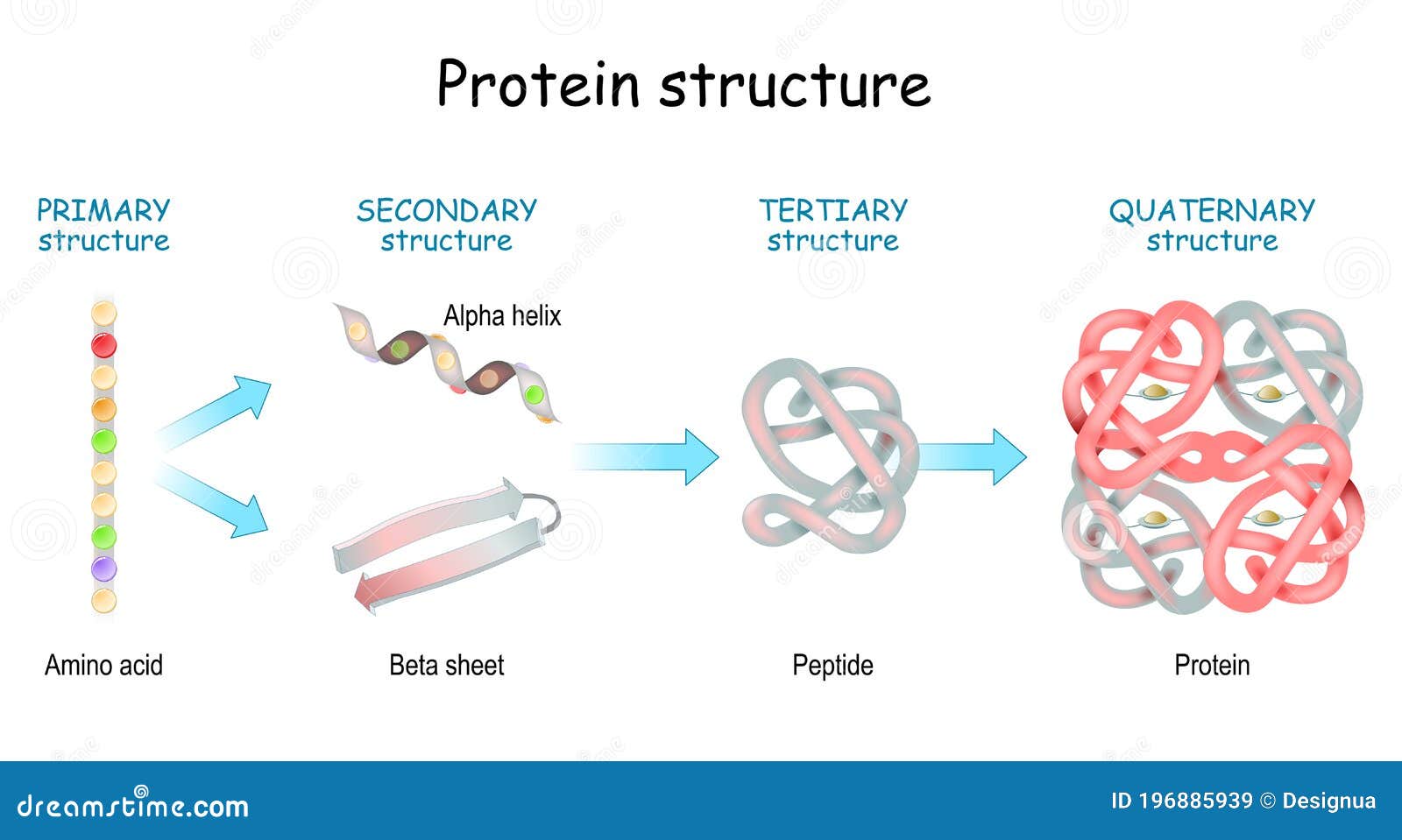
To form polypeptides and proteins, amino acids are joined together by peptide bonds, in which the amino or NH 2 of one amino acid bonds to the carboxyl (acid) or COOH group of another amino acid.All amino acids contain an amino or NH 2 group and a carboxyl (acid) or COOH group.There are 20 different amino acids commonly found in proteins and often 300 or more amino acids per protein molecule. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
